Oral Race, Open Questions
Week Of March 28 – April 3, 2026
The first oral non-peptide GLP-1 is here (approved). I go into what makes the orforglipron (Foundayo) approval significant, where it sits in a competitive oral landscape of 20+ programs, and the biological question that’s becoming central to the field: why does weight come back when you stop these drugs? Novo fired back with an indirect comparison claiming Wegovy pill beats Foundayo on both weight loss and tolerability. And Novo also registered two new amycretin (NNC0487-0111) trials this week, including a Phase 3 weight maintenance study - directly relevant to the regain question.
🔥 THIS WEEK’S KEY DEVELOPMENTS
FDA approves Lilly’s Foundayo (orforglipron), the first oral non-peptide GLP-1 for weight loss.
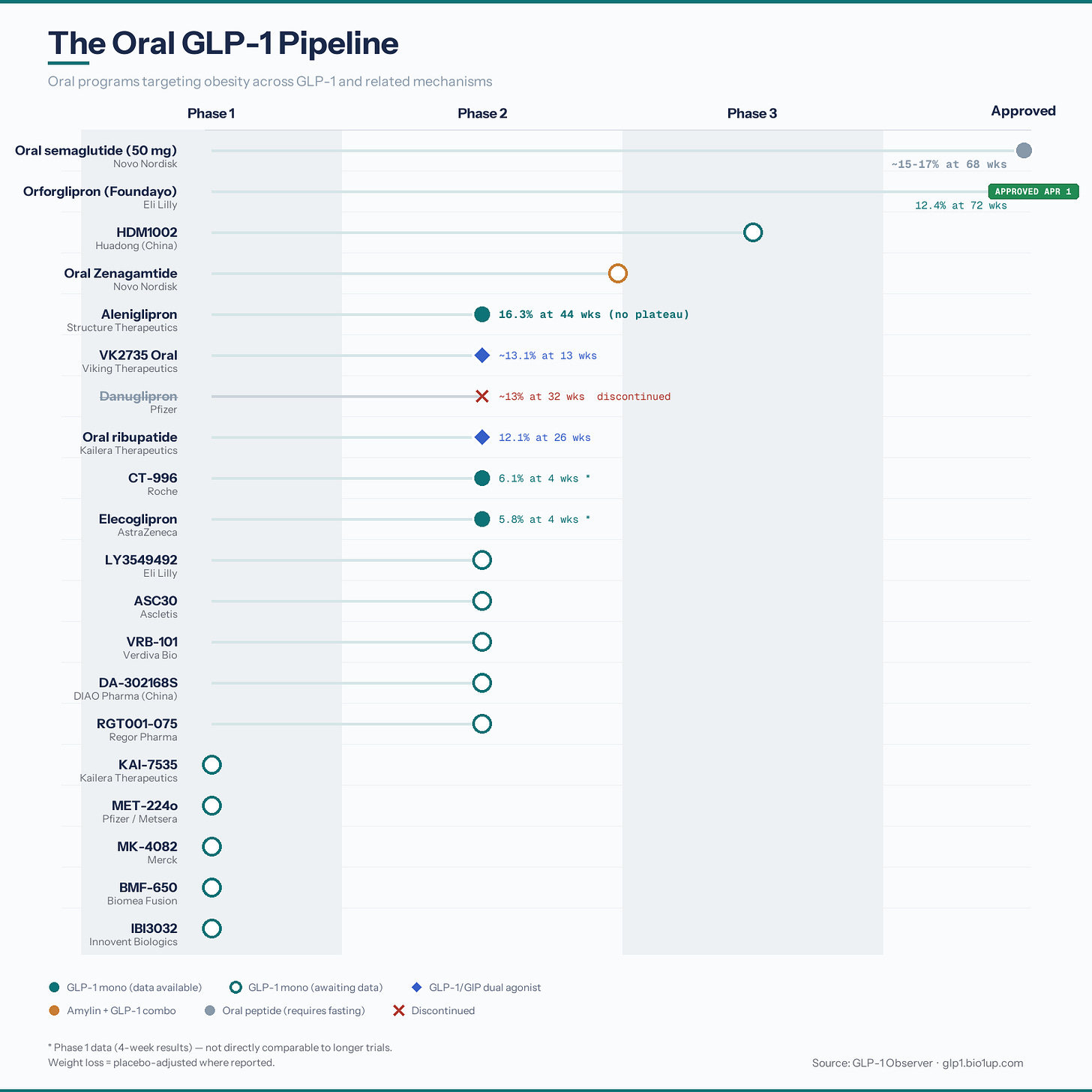
The FDA has approved Eli Lilly’s Foundayo (orforglipron), a once-daily oral non-peptide GLP-1 receptor agonist, for adults with obesity or overweight with weight-related medical problems. The approval was the first new molecular entity under the FDA’s National Priority Voucher program, completed in just 50 days. In the ATTAIN-1 trial, individuals taking the highest dose of Foundayo lost an average of 27.3 pounds (12.4%) compared to 2.2 pounds (0.9%) with placebo. Unlike oral semaglutide, the pill can be taken at any time of day without food or water restrictions.
FDA | Press | Trials: NCT05869903 | Mechanism: oral non-peptide GLP-1 receptor agonist
📰 PRESS
Novo Nordisk claims Wegovy pill beats orforglipron in indirect comparison.
Press | Mechanism: oral semaglutide
Taltz/Zepbound combo improves psoriatic arthritis and promotes weight loss.
Press | Trials: NCT06588296 | Mechanism: dual GLP-1/GIP receptor agonist
Lilly and Baseline investigate GLP-1s for substance use disorders.
Press | Mechanism: GLP-1 receptor agonist
Ambrosia raises $100M Series B for next-generation oral GLP-1s.
Press | Mechanism: oral small molecule GLP-1 receptor agonist
UK cost watchdog recommends Wegovy for cardiovascular risk reduction.
Press | Mechanism: GLP-1 receptor agonist
Kailera files for IPO to fund obesity drug ribupatide pipeline.
Press | Mechanism: GLP-1 receptor agonist
💡 TRIAL SPOTLIGHT
Educational spotlight selected by editors
Foundayo Arrives: Orforglipron Becomes the First Oral Non-Peptide GLP-1 - But the Race Is Just Starting
The FDA’s 50-day priority voucher approval of Eli Lilly’s orforglipron (Foundayo) on April 1, 2026, the fastest for a new molecular entity since 2002, marks a critical milestone as the first daily oral, non-peptide GLP-1 receptor agonist for weight management. Unlike peptide-based alternatives like oral semaglutide that require permeation enhancers and strict fasting, this small molecule partial agonist utilizes biased G protein signaling to enable convenient dosing at any time without food or water restrictions. The drug’s expansive clinical program provides a robust foundation for broad utilization, highlighted by the ATTAIN-1 obesity trial demonstrating 12.4% weight loss at 72 weeks, ATTAIN-MAINTAIN establishing a novel “injectable-to-oral step-down” paradigm, and ACHIEVE-3 proving superiority over oral semaglutide in diabetes. However, Foundayo’s launch merely fires the opening shot in a rapidly intensifying oral metabolic race, as prescribers now navigate between Lilly and Novo Nordisk’s offerings while tracking over 20 distinct programs advancing through a crowded pipeline. With Pfizer acquiring Metsera for $10 billion to reboot its pipeline and next-generation candidates like Structure Therapeutics’ aleniglipron already posting 16.3% placebo-adjusted weight loss with low discontinuation rates in Phase 2, the competitive landscape of oral incretins is poised for dramatic evolution over the next several years.
ClinicalTrials.gov: NCT05869903 (ATTAIN-1) | NCT06045221 (ACHIEVE-3) | Eli Lilly Press Release
🔬 MECHANISM EXPLAINED
Understanding the science behind the therapeutics
Why Weight Comes Back: The Biology of Regain After GLP-1 Therapy - And the Race to Solve It
Weight regain after stopping GLP-1 receptor agonists is a coordinated biological defense, not a return to old habits. Three converging forces drive it. First, hormonal counter-regulation: appetite hormones like ghrelin remain elevated and satiety signals like leptin and PYY remain suppressed for over a year after weight loss. GLP-1 drugs mask these changes during treatment but do not reset them, so the full force of this hormonal defense reasserts itself when the drug is removed. Second, metabolic adaptation compounds the problem. Total energy expenditure drops beyond what reduced body mass alone would predict, and because 25-40% of weight lost on GLP-1 drugs is lean muscle, patients end treatment with a lower metabolic rate but the same appetite drive. Third, emerging neuroscience reveals that the brain’s reward circuitry adapts to the drug itself. A 2025 Science paper found that ventral tegmental area (VTA) dopamine neurons recovered their responsiveness to palatable food even during ongoing semaglutide treatment, suggesting the hedonic system begins working around the drug before it is ever stopped.
The often-cited narrative that “you regain everything” is being revised. A March 2026 meta-regression in eClinicalMedicine found that regain follows a decelerating curve, plateauing at roughly 75% of weight lost. About 25% of the loss appears durably retained. Real-world data from a Cleveland Clinic study of 8,000 patients paints an even more favorable picture, largely because patients in practice restart medications rather than going cold turkey as withdrawal trial designs require. The field is now pivoting from maximizing acute weight loss to solving the maintenance problem. Lilly’s ATTAIN-MAINTAIN trial showed orforglipron maintained weight within 0.9 kg after switching from injectable semaglutide. Novo Nordisk registered AMAZE 12 this week, a Phase 3 amycretin trial specifically targeting weight maintenance. And non-incretin approaches like LX9851, which prevented weight regain after semaglutide discontinuation in preclinical studies, alongside muscle-preserving agents like bimagrumab, are attacking the problem from entirely different biological angles.
🆕 NEWLY REGISTERED TRIALS (4 in last week)
NNC0487-0111 Phase 1 trial assessing effects on food intake, appetite, and post-meal metabolism in people with obesity (Novo Nordisk, n=120)
[Weight Loss/Efficacy]
Trials: NCT07508020 | Mechanism: GLP-1 + amylin combination
MIST Phase 2 trial investigating a THR-β agonist in combination with semaglutide for metabolic disease (Eccogene, n=160)
[Weight Loss/Efficacy]
Trials: NCT07505303 | Mechanism: GLP-1 + THR-β combination
AMAZE 12 Phase 3 investigating NNC0487-0111 for weight loss maintenance in people with excess body weight (Novo Nordisk, n=600)
[Weight Loss/Efficacy]
Trials: NCT07503210 (AMAZE 12) | Mechanism: GLP-1 + amylin combination
RO7795081 Phase 1 investigating multiple oral doses in Chinese adults with obesity or overweight (Hoffmann-La Roche, n=30)
[Weight Loss/Efficacy | Oral Formulations]
Trials: NCT07499050 | Mechanism: GLP-1 receptor agonist
This newsletter compiles publicly available information from company press releases and ClinicalTrials.gov. Not investment advice.
Get in touch: Reply to this email, leave a comment on the post, or find me on X @GLP1observer. Explore the GLP-1 dashboard at glp1.bio1up.com.